Stock Products
RMLS provides you with your stem cell product research and translational needs. Learn more about our current offerings here.
UC - Mesenchymal Stem Cells
MSC Exosomes
MUSE Cells
Custom Exosomes and MSC
-
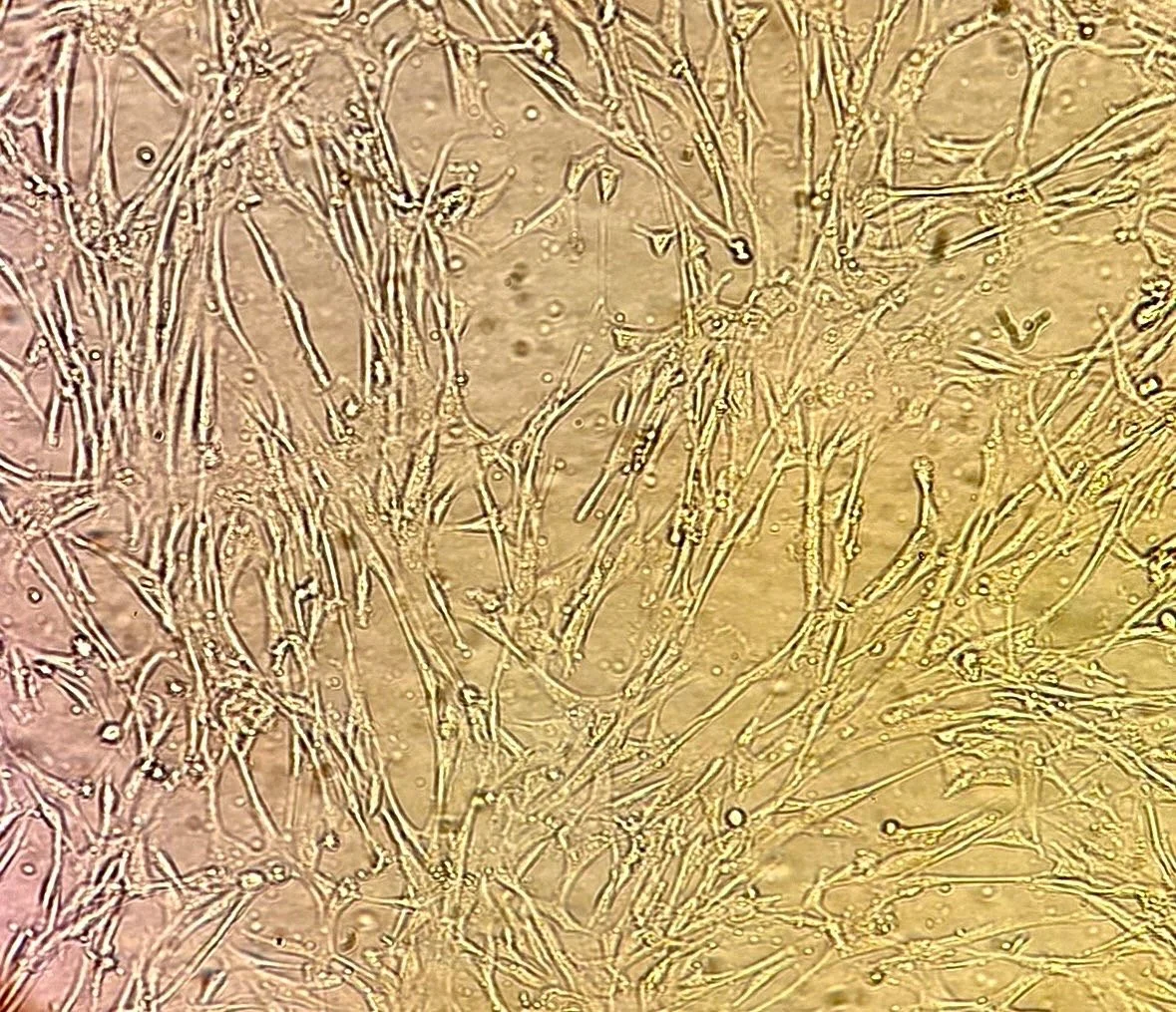
Umbilical cord–derived mesenchymal stem cells (UC-MSCs) are allogeneic stromal cells expanded from donated umbilical cord tissue under controlled manufacturing conditions. In this product format, UC-MSCs are produced at low passage to preserve a youthful phenotype and minimize culture-associated drift, then expanded in a 3D suspension system (e.g., on microcarriers in a closed, agitated bioreactor/spinner) to enable scalable, consistent growth while maintaining MSC identity and viability.
The final cell lot is released after comprehensive third-party microbiological safety testing, including:
• Sterility testing (no detectable bacterial or fungal contamination) using compendial/validated methods.
• Mycoplasma clearance testing (negative by validated assay) to address common culture-associated contaminants that may not be detected by standard sterility tests.
• Endotoxin testing to ensure endotoxin levels meet clinical-grade acceptance criteria (e.g., within applicable pharmacopeial/clinical limits for parenteral products), supporting suitability for downstream therapeutic applications.
Collectively, these controls support a UC-MSC preparation that is low-passage, 3D-expanded, and microbiologically qualified, with additional safeguards for mycoplasma and endotoxin beyond baseline sterility testing.
-
3D-cultured umbilical cord–derived mesenchymal stem cell (UC-MSC) exosomes are extracellular vesicles produced by UC-MSCs expanded under a near native cellular environment that supports consistent exosome production with a biologically relevant cargo profile.
These exosomes contain naturally occurring peptides, lipids, and RNAs associated with cell signaling, inflammation modulation, and tissue repair. UC-MSC–derived exosomes are nanoscale (approximately 30–150 nm), cell-free, and non-replicative, making them well suited for in-vitro and translational research applications, with potential for future clinical development.
Exosomes are produced under controlled laboratory conditions and are sterility tested using a third-party cGMP-compliant testing laboratory, consistent with the sterility testing approach used for UC-MSC cell products.
-
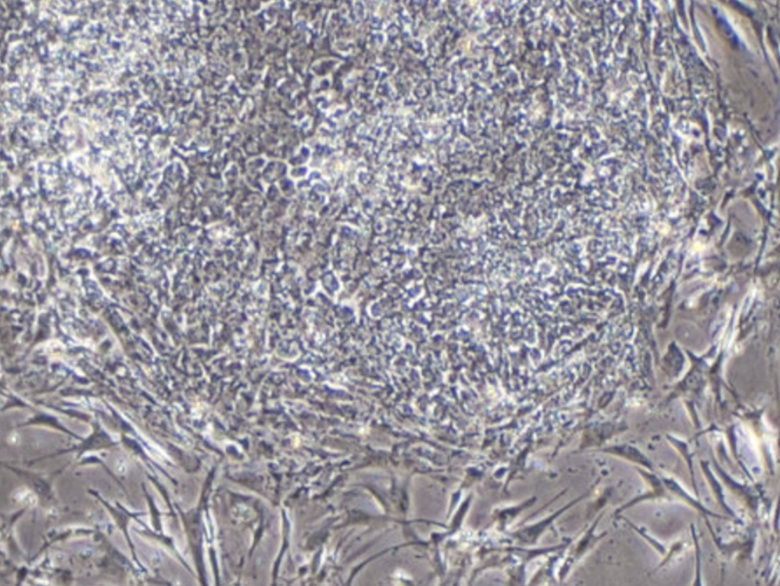
MUSE cells (Multilineage-Differentiating Stress-Enduring cells) are a rare, endogenous population of adult stem cells that combine broad, pluripotent-like differentiation potential with an exceptional safety profile. Naturally present within mesenchymal tissues, MUSE cells can differentiate into cell types from all three germ layers without genetic manipulation, while remaining non-tumorigenic and clinically stable. Their innate resistance to cellular stress, ability to home to sites of injury, and low immunogenicity make MUSE cells a powerful and practical platform for next-generation regenerative therapies—bridging the gap between pluripotency and safety.
-
RMLS offers cells and cell-derived exosomes produced under defined exposure conditions designed to promote anti-inflammatory profiles. Production incorporates carefully selected peptides and nutrient formulations with demonstrated relevance to immunomodulatory and anti-inflammatory pathways. These products are suitable for research, translational, and product development applications and can be produced at scale.
Contact RMLS to discuss current availability, production parameters, and opportunities for collaboration.